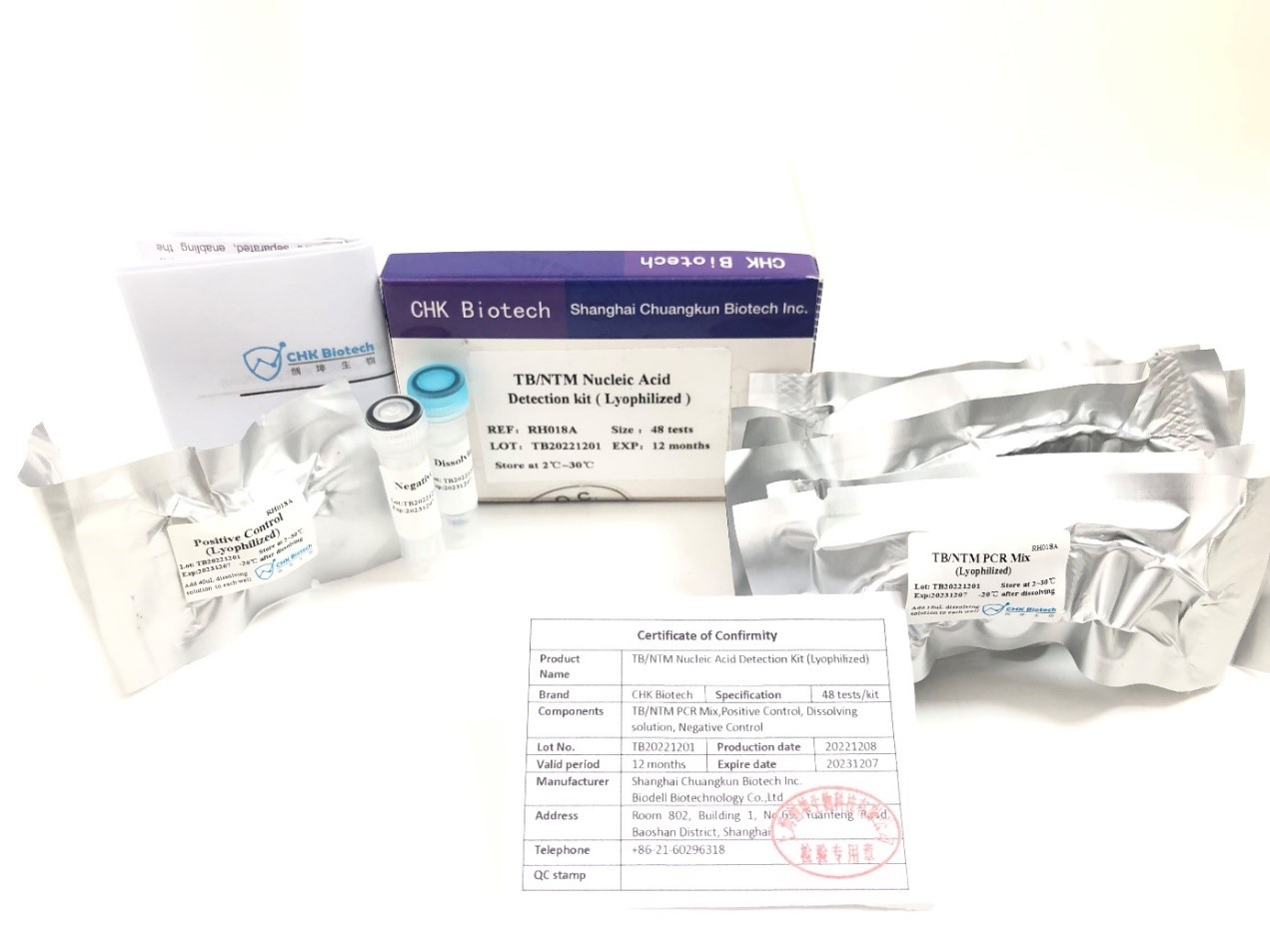
Recently, Shanghai Chuangkun Biotechnology Co., Ltd. obtained the second registration certificate of Indonesia FDA for TB/NTM Nucleic Acid detection kit(lyophilized), steped by the (12+3)type HPV detection PCR kit one month ago. which declared that Chuangkun Biotech’s products have been authorized by Indonesia FDA, improving self-developed products,and also providing strong support for Chuangkun Biotech to further expand the international market.
According to the World Health Organization’s Global TB Report 2022, an estimated 10.6 million people will develop TB in 2021.The increasing rate by 4.5% was more than the year 2020.Meanwhile 1.6 million people died of tuberculosis last year(including 6.7% were among people living with HIV). Geographically, most TB cases in 2021 were in the WHO regions of South-East Asia (45%), Africa (23%) and the Western Pacific (18%), with smaller shares in the Eastern Mediterranean (8.1%), the Americas (2.9%) and Europe (2.2%). Considering the COVID-19 Pandemic,tuberculosis prevention and controlling is more and more harshed for the Plan of Termination of Tuberculosis for WHO.
TB/NTM Nucleic Acid detection kit from CHUANGKUN Biotech is creatived by lyophilization procedure .The process will solve the long-term transportation disadvantages for the traditional PCR detection kit, and also brings excellent accuracy and high specificity results more over covenience for our uses.
Shanghai Chuangkun Biotechnology Co., Ltd. obtained the second registration certificate of Indonesia FDA for TB/NTM Nucleic Acid detection kit,indicates recognition by the Indonesia FDA. Chuangkun Biotech will be devoted to parctise in the regional tuberculosis prevention and controlling actively.
Post time: Jun-08-2023

 中文
中文